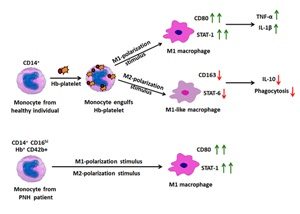
 The distinct response shown by different phenotypes of macrophages and monocytes under various clinical conditions have put the heterogeneity of these cells into focus of investigation for several diseases. Recently we have described that after engulfing Hb-activated platelets, classical monocytes differentiated into pro-inflammatory phenotypes, which were abundant in the circulation of PNH and SCD patients. Our current study shows that upon engulfment of Hb-activated platelets, monocytes differentiate into M1-macrophages under M1-polarization stimulus (GM-CSF, IFN-γ+LPS). When cells were grown under M2-polarization stimulus (M-CSF, IL-4+IL13), exhibited a M1-like phenotype, secreted elevated levels of pro-inflammatory cytokines including TNF-α and IL-1β, and displayed loss of the secretion of cytokine such as IL-10 and also phagocytic ability unlike the conventional M2 macrophages. Interestingly, when differentiated under the above polarization stimulus, monocytes from PNH patients expressed high levels of CD80 and phospho-STAT1, like M1 macrophages. Haemolytic mice also exhibited a gradual increase in monocyte-platelet aggregates in circulation and accumulation of CD80high macrophages in thioglycollate-induced inflamed peritoneum. The spleen of the mice was also populated by CD80high macrophages with compromised phagocytic capacity. Our findings suggest that the haemolytic environment and specifically the Hb-activated platelets, which are abundant in circulation during intravascular haemolysis, closely regulate monocyte differentiation. This article is protected by copyright. All rights reserved.
The distinct response shown by different phenotypes of macrophages and monocytes under various clinical conditions have put the heterogeneity of these cells into focus of investigation for several diseases. Recently we have described that after engulfing Hb-activated platelets, classical monocytes differentiated into pro-inflammatory phenotypes, which were abundant in the circulation of PNH and SCD patients. Our current study shows that upon engulfment of Hb-activated platelets, monocytes differentiate into M1-macrophages under M1-polarization stimulus (GM-CSF, IFN-γ+LPS). When cells were grown under M2-polarization stimulus (M-CSF, IL-4+IL13), exhibited a M1-like phenotype, secreted elevated levels of pro-inflammatory cytokines including TNF-α and IL-1β, and displayed loss of the secretion of cytokine such as IL-10 and also phagocytic ability unlike the conventional M2 macrophages. Interestingly, when differentiated under the above polarization stimulus, monocytes from PNH patients expressed high levels of CD80 and phospho-STAT1, like M1 macrophages. Haemolytic mice also exhibited a gradual increase in monocyte-platelet aggregates in circulation and accumulation of CD80high macrophages in thioglycollate-induced inflamed peritoneum. The spleen of the mice was also populated by CD80high macrophages with compromised phagocytic capacity. Our findings suggest that the haemolytic environment and specifically the Hb-activated platelets, which are abundant in circulation during intravascular haemolysis, closely regulate monocyte differentiation. This article is protected by copyright. All rights reserved.
For Full article: https://www.ncbi.nlm.nih.gov/pubmed/29677388


http://mewkid.net/order-amoxicillin/ – Amoxicillin Online Amoxicillin vni.nfmt.blogs.rcb.res.in.iwd.se http://mewkid.net/order-amoxicillin/
Buy Canine Prednisone Kamagra Oral Jelly Beograd buy antabuse online discount Propecia Hair Thickening Viagra Pro Generic Cialis Con Hipertension