Transposons are mobile genetic elements and their movement give rise to changes in the genome. These elements were discovered by Barbara McClintock and she was awarded the Nobel Prize for this discovery in 1983. The transposons usually code for enzymes known as transposases which are responsible for the movement of these mobile genetic elements. The activity of the transposase leads to excision of the transposon from its current and integration at another location in the genome. Transposition leads to changes in the genome and can give rise to cancer in animals or lead to the onset of drug resistance in pathogenic bacteria.
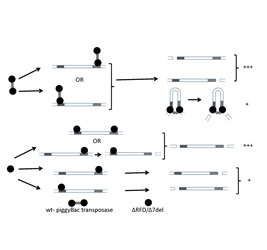
To understand how transposases function, we have studied the transposase from the eukaryotic piggyBac transposon. This transposon was first isolated from the Cabbage looper moth and has been used as a delivery vehicle for integration of the Yamanaka factors in the genome of cells to generate pleuripotent stem cells from differentiated cells. The piggyBac transposase binds to the terminal repeats present at the ends of the transposon. This is followed by excision of the transposon and release of the nucleoprotein complex. The complex translocates and is followed by integration of the transposon at the target site. We show that the RING-finger domain (RFD) present towards the C-terminus of the transposase is vital for dimerization of this enzyme. The deletion of the RFD or the last seven residues of the RFD results in monomeric protein that binds the terminal end of the transposon with nearly same affinity as wild type- piggyBac transposase. Surprisingly, the monomeric constructs exhibit more than two-fold enhancement in the excision activity of the enzyme. Overall, our studies suggest that dimerization attenuates the excision activity of the piggyBac transposase. This attribute of the piggyBac transposase may serve to prevent excessive transposition of the piggyBac transposon that may be catastrophic for the host cell.
For Full article: https://www.ncbi.nlm.nih.gov/pubmed/29750515


Cialis Por Vida Buy Cialis Ricetta Cialis Online canadian pharmacy cialis Zithromax Two First Day